Abstract Alzheimer's disease (AD) is considered as the most common cause of dementia in elderly population. While the exact mechanism of AD has not been discovered, hyperbolic [hyperbaric] oxygen therapy (HBOT) has been proven to be effective in the treatment of this...
Alzheimer’s Disease
Explore the latest research on the use of Hyperbaric Oxygen Therapy (HBOT) to treat Alzheimer’s Disease.
Extivita maintains an extensive publication database for Alzheimer’s Disease and various other indications with therapies such as Hyperbaric Oxygen Therapy, Neurofeedback, Nutritional IV Therapy, Infrared Sauna, and Pulsed Electromagnetic Field Therapy. Explore our database on Acne and Hyperbaric Oxygen Therapy aka, HBOT, below.
For a complete list of indications treated at Extivita, explore the conditions we treat.
Alzheimer’s disease: hyperbaric oxygen proposed as treatment in new study
Alzheimer’s disease, the most common form of dementia, has long been associated with a build-up of plaques (clumps of protein) in the brain. Scientists in Israel have shown that a type of oxygen therapy can stop new plaques forming and even remove existing plaques in...
Hyperbaric oxygen therapy alleviates vascular dysfunction and amyloid burden in an Alzheimer’s disease mouse model and in elderly patients
Abstract Vascular dysfunction is entwined with aging and in the pathogenesis of Alzheimer's disease (AD) and contributes to reduced cerebral blood flow (CBF) and consequently, hypoxia. Hyperbaric oxygen therapy (HBOT) is in clinical use for a wide range of medical...
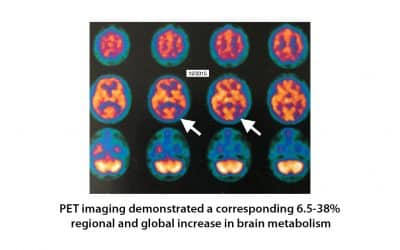
Hyperbaric oxygen therapy for Alzheimer’s dementia with positron emission tomography imaging: a case report.
A 58-year-old female was diagnosed with Alzheimer’s dementia (AD) which was rapidly progressive in the 8 months prior to initiation of hyperbaric oxygen therapy (HBOT). Fluorodeoxyglucose (FDG) positron emission tomography (PET) brain imaging demonstrated global and typical metabolic deficits in AD (posterior temporal-parietal watershed and cingulate areas). An 8-week course of HBOT reversed the patient’s symptomatic decline. Repeat PET imaging demonstrated a corresponding 6.5-38% regional and global increase in brain metabolism, including increased metabolism in the typical AD diagnostic areas of the brain.
The treatment of cognitive dysfunction in dementia: a multiple treatments meta-analysis.
No cure is currently available for dementia; however, various treatments and interventions have been reported to be effective. The factors influencing the efficacy of dementia treatment have not been comprehensively evaluated. This study evaluated the factors influencing treatment effects on cognitive dysfunction in dementia by comparing the results obtained from a meta-analysis based on meta-regression. The most effective intervention for dementia available is symptomatic treatment for vascular dementia. Antipsychotic treatment for dementia alleviates cognitive dysfunction less effectively than does symptomatic treatment. Alternative therapies are also effective at present. Further research on causes and very early diagnosis of Alzheimer disease is warranted.
Hyperbaric Oxygen Therapy for Alzheimer’s Disease.
Recent studies indicate that hyperbaric oxygen therapy (HBOT), a well-established therapy for decompression illness, could be a potential therapy for Alzheimer’s disease (AD). However, due to oxygen toxicity i.e., increased oxidative stress implicated in HBOT, the risk and benefit of HBOT for AD patients need to be further assessed clinically.
Nicotinamide riboside restores cognition through an upregulation of proliferator-activated receptor-γ coactivator 1α regulated β-secretase 1 degradation and mitochondrial gene expression in Alzheimer’s mouse models
Abstract Nicotinamide adenine dinucleotide (NAD)(+), a coenzyme involved in redox activities in the mitochondrial electron transport chain, has been identified as a key regulator of the lifespan-extending effects, and the activation of NAD(+) expression has been...
NAD+ biosynthesis, aging, and disease
Abstract Nicotinamide adenine dinucleotide (NAD+) biosynthesis and its regulation have recently been attracting markedly increasing interest. Aging is marked by a systemic decrease in NAD+ across multiple tissues. The dysfunction of NAD+ biosynthesis plays a critical...
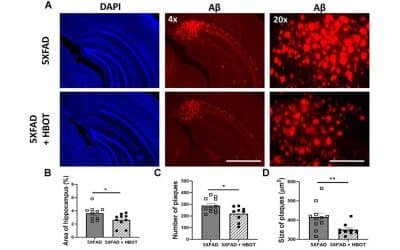
Hyperbaric oxygen therapy ameliorates pathophysiology of 3xTg-AD mouse model by attenuating neuroinflammation.
There is a real need for new interventions for Alzheimer’s disease (AD). Hyperbaric oxygen therapy (HBOT), the medical administration of 100% oxygen at conditions greater than 1 atmosphere absolute, has been used successfully to treat several neurological conditions, but its effects on AD pathology have never been thoroughly examined. Therefore, we exposed old triple-transgenic (3xTg) and non-transgenic mice to HBOT followed by behavioral, histological, and biochemical analyses. HBOT attenuated neuroinflammatory processes by reducing astrogliosis, microgliosis, and the secretion of proinflammatory cytokines (IL-1β and TNFα) and increasing expression of scavenger receptor A, arginase1, and antiinflammatory cytokines (IL-4 and IL-10). Moreover, HBOT reduced hypoxia, amyloid burden, and tau phosphorylation in 3xTg mice and ameliorated their behavioral deficits. Therefore, we suggest that HBOT has multifaceted effects that reduce AD pathologies, even in old mice. Given that HBOT is used in the clinic to treat various indications, including neurological conditions, these results suggest HBOT as a novel therapeutic intervention for AD.